

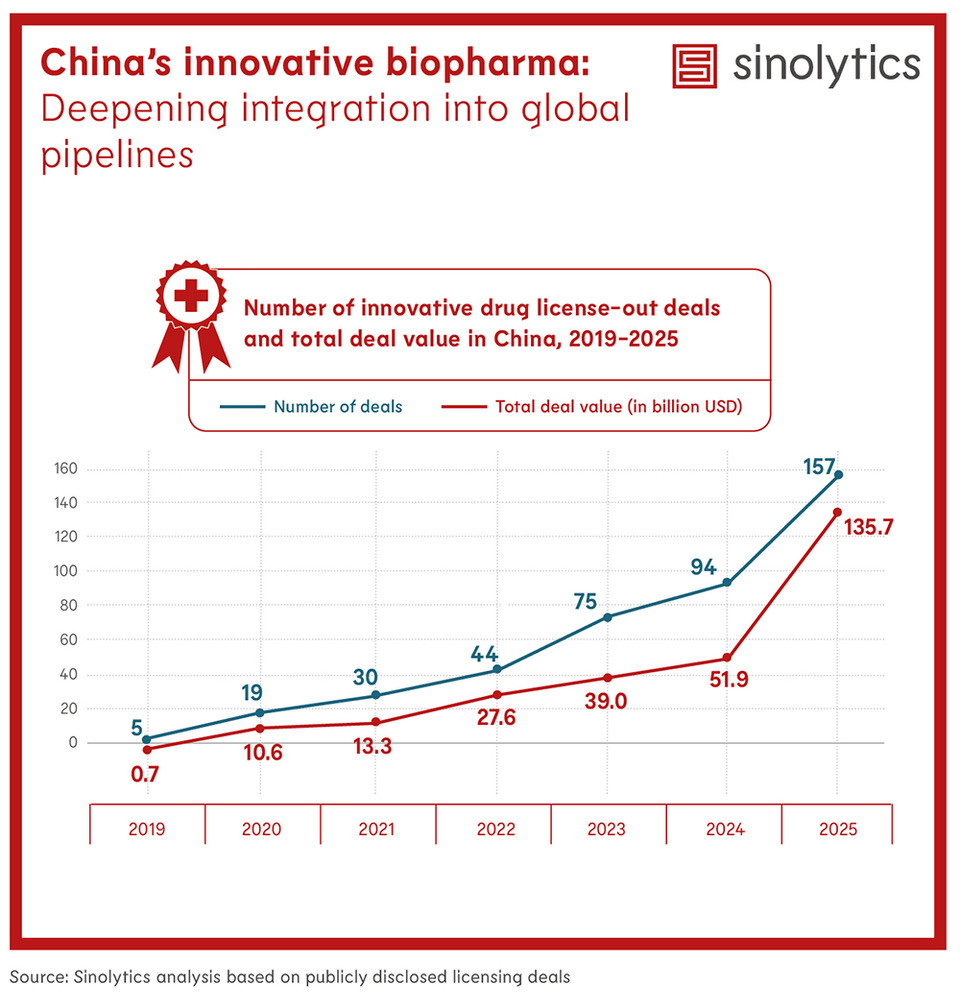
Cross-border licensing activity for Chinese innovative drugs has expanded significantly. In 2025, China’s innovative drug license-out activity increased sharply. The number of deals rose from 94 in 2024 to 157 in 2025, while disclosed deal value increased from about USD 51.9 billion to around USD 135.7 billion. This trend indicates that Chinese pharmaceutical companies are playing a growing role in supplying innovative drug assets to global pharmaceutical pipelines.
 Global pharma takes notice
Global pharma takes noticeOne driver behind the rise in licensing deals is that multinational pharmaceutical companies are actively seeking external drug candidates as many of their existing products approach patent expiration. Chinese biotech assets, particularly in areas such as oncology and metabolic diseases, are increasingly considered in global business development strategies. As of early 2026, this trend appears to be continuing.
Policy support in China is gradually expanding from R&D incentives to commercialization and payment systems. In March 2025, China’s State Council issued a policy supporting the high-quality development of innovative drugs, emphasizing support across the entire innovation chain. Later in 2025, China’s drug regulator (NMPA) further optimized the clinical trial review process for innovative drugs. The 2026 Government Work Report also highlighted the development of commercial health insurance and the promotion of innovative drugs and medical devices, indicating that policy support is increasingly extending from R&D to commercialization and payment mechanisms.
Geopolitical factors have not stopped cross-border biopharmaceutical cooperation, but the policy environment is becoming more complex. Several large licensing deals between Chinese and multinational pharmaceutical companies were still announced in early 2026, including a partnership between Sino Biopharm and Sanofi, indicating that cross-border cooperation continues. At the same time, the U.S. signed the FY2026 National Defense Authorization Act (NDAA) in December 2025, which includes BIOSECURE provisions restricting federally funded entities from using biotechnology equipment or services from “biotechnology companies of concern.” Earlier drafts of the legislation in 2024 explicitly named several Chinese companies, but the final law applies restrictions through a broader designation mechanism. As a result, cross-border cooperation is likely to continue but with greater emphasis on compliance and risk management.
This Sinolytics.Radar is a China-specific follow-on to our broader analysis of pharmaceuticals and geopolitics in 2026.